Typically, you need at least 8 steps to determine the electron configuration, starting with finding the atomic number by looking at the list of orbitals and understanding the notation. Therefore, to shorten the configuration for silicon she wrote Ne 3s2 3p2. This electron configuration calculator will instantly show you the distribution of electrons in the orbitals of any periodic element you choose. 
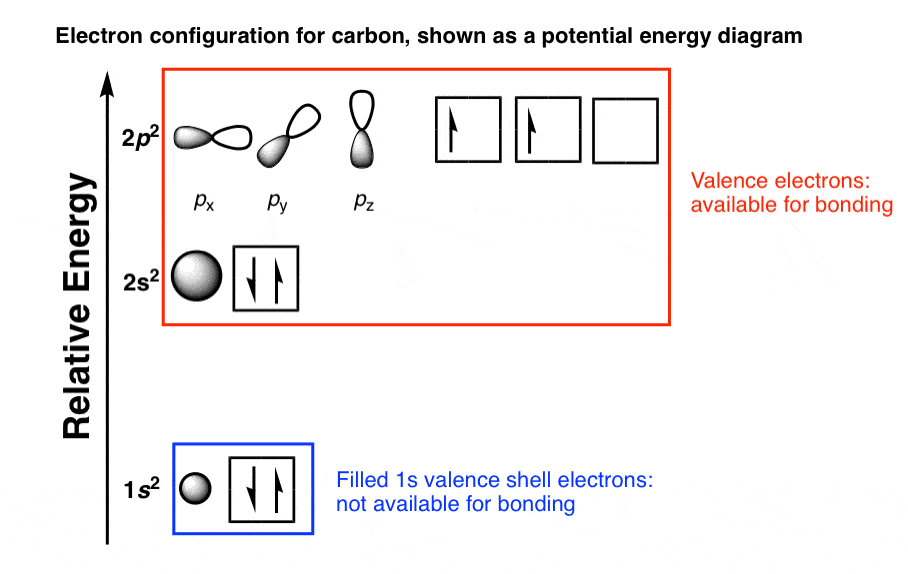
In writing the electron configuration for carbon the first two electrons will go in the 1s orbital. However, the first part of this long expression, 1s2 2s2 2s6, is the electron configuration of neon. Carbon is the sixth element with a total of 6 electrons. Written in full this is 1s2 2s2 2s6 3s2 3p2. Using the same method, calcium has 2 valence electrons. What she was doing was writing the electron configuration of silicon. For the element aluminum, above, we see 2 electrons in the 3s orbital and 1 electron in the 3p orbital, so aluminum has a total of 3 valence electrons. These electrons are important because they are the ones that are gained, lost or shared in chemical reactions. We typically do not consider “d” electrons as valence electrons and therefore a more specific definition is needed: valence electrons are those electrons in the highest principal energy level. In many cases, “d” electrons will be present after the last noble gas, as in the element manganese : 4s 23d 5. Enter the full electron configurations for each of the following elements. Explanation: Carbon with atomic number 6, has an electronic configuration of 1s2. The electron configuration of carbon is 1s22s22p2 and it reveals that carbon has 4 valence electrons in the 2 s and 2 p orbitals. Electron configuration can be done in two ways. Since it has four valence electrons it will form four covalent bonds. The electron configuration of carbon is 1s 2 2s 2 2p 2, if the electron arrangement is through orbitals. In the space below, write the abbreviated electron configurations of the following. The arrangement of electrons in carbon in specific rules in different orbits and orbitals is called the electron configuration of carbon. The electrons written after the noble gas in brackets are called valence electrons. 15 2520353048 3d 4p 58 4d15p 6s 4 f 50 6p 75 5f5. These electrons do not participate in chemical reactions. 
Those electrons in the brackets are called core electrons. Typically, the sharing of electrons is observed to complete the valance shells by. Valence Electrons-This notation for writing electron configurations helps us to highlight 2 different types of electrons in the atom. Carbon group the group 14 elements have four electrons in the outer shell. Electron Configuration Oxidation States Plot Electronegativity (Pauling. We know from the periodic table that copper is in group 11, so this agrees with the result of this electron configuration calculator.We may NOT use any element in the brackets, only noble gases. For example, Carbon had an atomic weight of 12.00 in 1902 but today it is. Let us look at the electron configuration for helium, which is 1 s 2 1 4s^1 3 d 10 4 s 1. The electron configuration for the first 10 elements. The electron configuration is responsible for many physical and chemical properties of an element. Electron Configurations are an organized means of documenting the placement of electrons based upon the energy levels and orbitals groupings of the periodic table. Carbon, with symbol C, is a non-metallic and tetravalent chemical element, which means that it has four electrons to form chemical bonds. Ground state means that the atom has the lowest energy allowed. Electron configuration notation provides us with information about the basic energy levels and sublevels that electrons occupy. The isotope shown here is carbon-12, with a nucleus of 6 protons (red) and 6 neutrons (blue). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
